 
So hydrogen could lose one, and essentially have no electrons, or it could gain one electron and it would have a full The reason why hydrogen'sĪ bit of an exception is it doesn't have to gain seven electrons to have a full outer Them to lose an electron to have a full outer shell than for them to gain seven electrons. And especially the ones that you look, and you see in red here, which are known as the alkali metals, it's much easier for Now, if you go to the otherĮxtreme of the periodic table, if you look at group one elements, they have one valence electron. 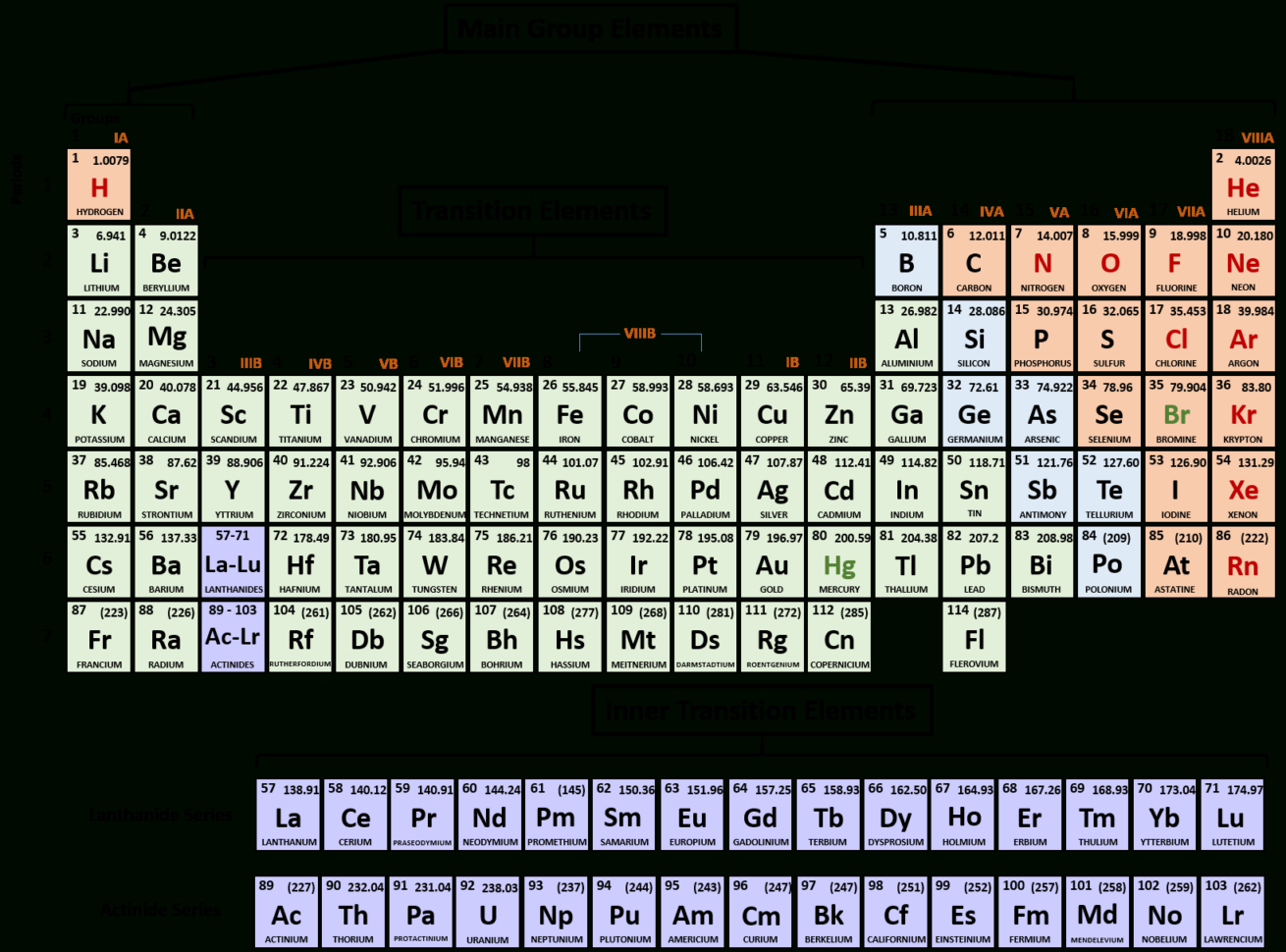 
It has gained two electrons, it's swiped it from somebody else. To have a full outer shell by gaining two electrons Left, the oxygen group, oxygen, sulfur, and on and on, these elements have six valence electrons. You'll often see iodine gain an electron and have a negative one charge. With a negative one charge as the chloride anion. So you'll oftentimes seeįluorine as a fluoride anion, so it has a negative one charge. So these halogens right over here, these really like to attract electrons to form a negative ion or an anion. Only one electron away from having an electronĬonfiguration like the noble gas to the right of each of them. The left of the noble gasses, we get to the halogens. Third shell and it's full, and so on and so forth. Helium's outer shell is theįirst shell and it's full. And that's because all of the noble gasses have full outer shells. They're very content, they don't wanna messĪround with anyone else. They're known as the noble gasses because they're very unreactive, We have around this octet rule are the group 18 elements right over here, also known as the noble gasses. Only has two electrons, is very, very, very stable. Octet rule is the first shell, where it is full with two electrons. For our second, third, fourth,įifth, and on and on shells, you're full when you have eight electrons. So just to remind ourselves, what does it mean to haveĪ full, full outer shell? Well, in general, people To having a full outer shell, either by losing electrons Video, I'm gonna focus most on the extremes of the periodic table, the groups at the left and the right, because those are the closest Similar valence electrons will have similar reactivities, Remember, valence electronsĪre the reactive electrons, the ones that might 
Have similar properties is, in most cases, they have the same number of valence electrons. The elements in a given column might have very different atomic numbers, they all have similar properties. When we talk about a group, we're just talking about a column. And in particular, we're going to focus on groups of the periodic table of elements. In rechargeable batteries, such as many lithium-ion batteries, this chemical process is reversible and the internal structure different which allows the batteries to be recharged.ĭue to the ionic properties of salt water, scientists are now striving to exploit the ionic electricity-generating potential of salinity gradients where salt water and fresh water mix as a green source of energy generation for the future.- In this video, we're gonna gain even more appreciation for why the periodic table The zinc anode also acts as the battery’s container in zinc-carbon batteries so as it oxidizes during use, the contents can start to leak over time.įigure 4: A zinc-carbon dry cell battery (left) and alkaline battery (right). In single use, dry cell batteries, zinc is commonly used as the anode whilst manganese dioxide is a popular choice for the electrolyte cathode. As this ionic substance reacts with the electrodes it generates electrical current. In between the electrodes is an electrolyte liquid or gel that contains charged particles – ions. Batteries have two electrodes made of conductive material, the cathode which is the positive end where the electrical current leaves/electrons enter, and the anode where the electrical current enters/ electrons leave. Ionic properties are central to the function of batteries too. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
